Difference between revisions of "The Nexian DMT Handbook"
(→"The Machine": info about CuO) |
Stoneyjonez (Talk | contribs) (→Storage Concerns) |
||
| Line 494: | Line 494: | ||
==='''Storage Concerns'''=== | ==='''Storage Concerns'''=== | ||
| + | DMT should be stored in a cool and dry place, preferably in a sealed container. | ||
==='''Discussion of Oxidization'''=== | ==='''Discussion of Oxidization'''=== | ||
Revision as of 09:55, 22 May 2009
| Note: | This page is a work in progress -- its content throughout is not yet complete. |
The production and use of DMT (N,N-dimethyltryptamine), otherwise known as "Spice", is a practice that resonates strongly with the complementary qualities of ancient shamanic and alchemical spiritual practice as well as contemporary DIY (Do It Yourself) ethic. The production of spice is a discipline unlike that of most other commonly manufactured drugs, as it is not as well suited for bulk-production nor production for the purpose of sale as most well-known and intensively manufactured substances. As such, its use is generally inseparable from its production in practice and in spirit.
The production of DMT most commonly entails its extraction from botanical sources and only very rarely entails its synthesis. In this way, its production still strongly resembles its more ancient preparations by manner of brewing, a simple form of aqueous extraction still commonly performed to this day. This is the simplest and most readily administered form of extract if used as a component of a harmaloid-based preparation or -huasca brew.
Please take the time to seek further elaboration at the following resources:
Contents
Source Selection
DMT, its analogues, and other related alkaloids can be found in a wide variety of lifeforms, varying from trace amounts to considerable amounts. It is impossible, therefore, to include all of the sources from which DMT can be extracted, so the following discussion will focus primarily on the most commonly used and significant botanical sources.
Botanical Considerations
Several species of plants contain a variety of constituents apart from DMT. This consideration is of the utmost importance when selecting the source plant from which an extraction is to be performed, as it may become the determining factor in the material requirements of the extraction process. Some plants may even contain toxic alkaloids, so thorough research must be conducted prior to selection, extraction, and administration.
Common Botanical Sources
| Mimosa hostilis |
|
|---|---|
|
| Mimosa ophthalmocentra |
|
|---|---|
|
1,6% DMT in the inner rootbark [1] |
| Acacia obtusifolia |
|
|---|---|
|
See also:
Methods of Refinery
Before extracting alkaloids from the variety of plants named above, one generally needs to clean and prepare the plant source, to include washing, pulverization, or any other necessary pre-treatment.
Defatting Concerns
Vendor Considerations
Considerations Regarding Cultivation
Extraction
Extraction generally refers to the process of isolating a product from a source. The basic idea is to utilize the unique properties of the product—whether reactive, electromagnetic, or otherwise structural—to draw it out of the source and into a target solvent. To accomplish this the product must either be naturally soluble in the solvent or must undergo reaction to increase its solubility. The difference between a high and low yield is firstly determined by how much more soluble the product is in the target solvent, than in its source material or solution, and secondly by how thoroughly the target solvent is mechanically brought into contact with the target solvent.
See also:
STB Techniques
Considerations:
- The use of "Straight To Base" techniques requires little experience or technical know-how for beginners to approach extraction techniques. STB is best-suited for quick, non-labor-intensive, crude bulk extractions. It requires no straining or cooking but requires time for soaking and separation. STB tends to yield a greater array of botanical impurities due to its lack of straining and defatting. These techniques do however enable a more thorough exhaustion of product from the material. This technique is ideal for shredded material that requires little or no defatting.
Overview of Materials and Methods
| Materials Required |
|---|
Methods:
Material Preparation
Extraction Procedure
Further Elaboration and Technical Support
A/B Techniques
Considerations:
- The use of Acid/Base techniques implies the use of "acid-cooking" the source material, straining it, and basifying the resulting strained solution. The use of an initial acid extraction facilitates the implementation of a defatting phase and generally yields a product more devoid of botanical impurities. This technique is ideal for any material that requires defatting, though defatting may not be necessary, depending on the intended method of crystallization.
Overview of Materials and Methods
| Materials Required |
|---|
Methods:
Material Preparation
Extraction Procedure
Further Elaboration and Technical Support
Dry Techniques
Dry techniques (drytek) evolved from and are ideally intended for the implementation of the FASA method of crystallization and serve as the only techniques able to implement acetone as an extraction solvent. Acetone is generally favored for its ability to extract a notably broad range of active products.
Considerations:
- The use of dry techniques requires fewer and less toxic materials than the techniques that employ aqueous phases and separatory methods. The materials required are generally of a more household nature. They are most effectively applied to powdered botanical material. Acetone is, however, completely water miscible, so proper drying procedures are of the utmost importance. This technique may or may not require the defatting of botanical materials, depending on the intended method of crystallization. Dry techniques are the youngest of the current extraction techniques though apparently sound in theory and in practice.
Overview of Materials and Methods
Material Considerations:
- Acetone can be purchased at hardware stores but should be confirmed as pure acetone prior to purchase. Note that almost all acetone can contain up to 5% water contamination, depending on time and shelving conditions.
- In order to be utilized for extraction, sodium bicarbonate must undergo conversion to sodium carbonate.
- Lime is often found difficult to decant acetone off of and also difficult to filter out of acetone, whereas sodium carbonate is generally found more agreeable for both.
- Distilled water is preferable, as tap water almost always contains impurities that can potentially tamper with resulting yields.
- With few exceptions, the source material should be completely pulverized to a powder consistency before use, as this technique's choice of reagents are not quite capable of penetrating cell structure.
Methods:
- Extractions by dry techniques are characterized by the lack of a traditional aqueous phase in the extraction process, and instead, opting for basification within a paste which is followed by chemically drying the paste with desiccant. The process does not make use of separatory methods, and instead is characterized by it's use of dry-washing, decanting and non-intensive filtering methods. Certain materials must be rendered anhydrous prior to use.
Material Preparation
Extraction Procedure
- Mix the intended base with the powdered source material at a ratio between 1:2 and 1:1.
- The product remains in its natural salt form which is generally considered to be quite free from the botanical cell structure in powdered material.
- Add only enough water to thoroughly moisten the mixture to the consistency of a paste while stirring to ensure the consistency of the mixture.
- Although this is not generally considered a traditional aqueous phase in that it is not a solution, it is an aqueous phase in that it is excessively hydrated and sufficiently aqueous to facilitate reaction.
- Allow adequate time to soak in order for reaction to occur.
- The acid component of salt-form product undergoes reaction with the base, effectively neutralizing the acid and freeing the product in its pure alkaloid form, or freebase.
- Stir in anhydrous magnesium sulfate until thoroughly dry.
- The magnesium sulfate acts as a desiccant, and that this is performed in order to prevent water contamination of the acetone.
- Add an excess of anhydrous acetone and stir thoroughly, allotting adequate time and stirring for thorough dissolution of the product into the acetone.
- The more contact allotted between the product and the acetone, the greater the saturation.
- Decant and/or filter acetone and collect, being careful not to allow any particulates into the collection vessel.
- The bases used should not harm the quality of the product, but may interfere with the accuracy of weight.
- Repeat steps 5-6 with fresh acetone until material is exhausted to satisfaction.
- Three washes is generally considered sufficient.
Further Elaboration and Technical Support
Crystallization
Crystallization is the process by which a product is isolated from a solvent. This is accomplished by either allowing the solvent to completely evaporate or by causing a precipitation to occur within the solvent, which can then be isolated from the solvent by several methods and then dried of any residual solvent.
Evaporation
In extraction, evaporation is the process by which a solvent disperses from its liquid form into the air as a vapor and a gas. When this occurs, the less volatile constituents of the solvent solution are left behind, and as such, it is a common method of isolating solutes from solvent.
Considerations:
- Many common solvents contain impurities which may not be quite as volatile as the pure solvent and may leave these impurities behind as a residue.
- Solvents often emit fumes and odors which may be hazardous to health, flammable, or may alarm those within proximity of the odor.
- Some solvents may require an excessive length of time to evaporate.
- Some solvents may absorb ambient moisture, resulting in a less expedient evaporation.
- Excessive air flow may cause the oxidization of the product.
- Solvent may become Trapped within the crystal structure of the product, resulting in a less solid and less pure product.
Freeze Precipitation
Freeze precipitation is the process by which product is isolated from a solvent through a decrease in solubility achieved by lowering the temperature of the solvent. This process generally relies on the solvent being completely saturated or super-saturated with product. Freeze precipitation is generally the fastest method by which product can be isolated immediately following extraction, but it relies on the use of only very specific solvents. This method is preferably used in conjunction with A/B and STB techniques.
Salting Methods
Salting is the process by which freebase DMT is reacted with an acid to create a salt form which is generally water-soluble. The natural form of DMT in botanical sources tends to be a salt-form, thus facilitating the simple aqueous extraction used to prepare DMT-containing brews. It is quite common to perform aqueous acid extractions from the material, however—whether for the purposes of a brew or for A/B extraction. The salt-form itself rarely lends itself to proper crystallization and usually can only be isolated as an oil unless very specific methods and materials are employed.
The FASA Method
| Materials Required | |
|---|---|
| Source Material: |
|
Solvents:
|
|
Reagents/Desiccants:
|
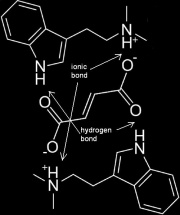
The FASA, or fumaric acid saturated acetone, method is a method employed to render DMT Fumarate.
Considerations:
- DMT Fumarate is reportedly quite stable and resistant to oxidization or other forms of degradation. It is notably resistant to heat, and as such is able to withstand low-temperature oven-drying. Certain other related compounds, such as jungle-spice and bufotenine are also able to crystallize as a fumarate. Defatting is not required prior to employing FASA methods, as oils and most other impurities should not interfere with this method's procedure or the yield
- Because DMT Fumarate is water-soluble, it is also well-suited for oral administration in conjunction with harmaloids, either mixed into a beverage or encapsulated.
Methods:
- The FASA method employs the firstly, the solubility of fumaric acid in acetone, and secondly, solubility of freebase DMT in acetone, and thirdly, the insolubility of DMT Fumarate in acetone or the non-polar solvents commonly utilized for extraction. The solubility of both DMT and fumaric acid in acetone facilitates their reaction to produce a crystalline DMT salt which is completely insoluble in acetone or non-polar solvents.
| Rendering Crystalline DMT Fumarate | |
|---|---|
|
Further Elaboration and Technical Support
Purification
The purification of DMT product has several purposes and is accomplished by several different methods, but all of them essentially involve the washing of product in some way or another. Purification either involves the isolation of product from unwanted impurities from the plant source or from the process of extraction, or it involves the isolation of product from active impurities which may or may not be collected after isolation.
Recrystallization
The general purpose of recrystallization is to crystallize the product in a fresh solvent after it has already been isolated from a solvent containing a considerable amount of impurities. This is meant to lessen the interference of impurities on the process of crystallization. Often this process results in more well-formed crystals with less discoloration. The advantage of this method of purification is that the solvent choice for recrystalliztion may be different and more suitable than that chosen for extraction.
Washing
The purpose of washing is to disperse impurities off of the product or out of a solution containing the product and into an intermediate solvent.
Alkaline Solution Washing of Inactive Impurities
Most of the impurities that plague yields tend to be quite soluble in both alkaline aqueous solutions and non-polar solvents. To remove these impurities, an imbalance in equilibrium must be created between these two types of solutions, causing the impurities to disperse into a disposable solution from the solution containing the product.
Solvent Washing and Isolation of Active Impurities
Active impurities require a slightly different method of isolation for purification and generally rely strictly on differences of their solubility or insolubility in specific solvents. Often, reactions are required in order to create these differences, as the products tend to exhibit very similar properties.
Freebase Conversion from Salt
The methods used for converting crystalline salt-form DMT into freebase are not dissimilar from those used in extraction. The only significant difference between the processes is that the conversion involves far fewer impurities and less material than the extraction. Because of of this and the fact that it involves the isolation of the product from an acid, the conversion acts as a sort of purification method.
STB Conversion
This conversion is simplistic in that it almost exactly resembles the methods used in STB Techniques, though it is significantly simpler in that it involves less material, fewer impurities, and does not require a strong base.
| Materials Required | |
|---|---|
| Source Material: |
|
Solvents:
|
|
Reagents/Desiccants:
|
Procedure:
- Dissolve DMT Fumarate in an adequate amount of water.
- No reaction occurs at this point.
- Add a concentrated solution of weak base until total precipitation is observed by the cloudiness of the solution.
- The fumaric acid undergoes reaction with the base, effectively neutralizing the acid and freeing the product in its pure alkaloid form, or freebase.
- Stir in NPS thoroughly and allow to separate from the aqueous solution.
- The more contact allotted between the product and the NPS, the greater the saturation.
- Collect the top layer of NPS, being careful not to allow any aqueous contamination.
- Aqueous contamination may result in an impure product or may disrupt subsequent crystallization.
- Repeat steps 3-4 with fresh or unsaturated NPS until the aqueous solution is exhausted to satisfaction.
- Three washes is generally considered sufficient.
- See Crystallization in order to render crystalline freebase product.
Drytek Conversion
This conversion is preferred for it's lack of need for separatory methods and for it's notably dry quality which facilitates the use of acetone. This method of conversion evolved out of the FASA method and is characteristically identical to Drytek extraction.
| Materials Required | |
|---|---|
| Source Material: |
|
Solvents:
|
|
Reagents/Desiccants:
|
Procedure:
- Mix DMT Fumarate with the intended base at ratio of about 1:1.
- No reaction occurs at this point.
- Add only enough water to thoroughly hydrate the mixture to the consistency of a paste while stirring to ensure the consistency of the mixture.
- The fumaric acid undergoes reaction with the base, effectively neutralizing the acid and freeing the product in its pure alkaloid form, or freebase.
- Stir in anhydrous magnesium sulfate until thoroughly dry.
- The magnesium sulfate acts as a desiccant, and is performed in order to prevent water contamination of the acetone.
- Add an excess of anhydrous acetone and stir thoroughly, allotting adequate time and stirring for thorough dissolution of the product into the acetone.
- The more contact allotted between the product and the acetone, the greater the saturation.
- Decant and/or filter acetone and collect, being careful not to allow any particulates into the collection vessel.
- The bases used should not harm the quality of the product, but may interfere with the accuracy of weight.
- Repeat steps 5-6 with fresh acetone until material is exhausted to satisfaction.
- Three washes is generally considered sufficient.
- See Crystallization in order to render crystalline freebase product.
Further Elaboration and Technical Support
Administration
Leaf Enhancement
Vaporization
The vaporization method of administration pertains to the use of pure freebase product with no additional material. This method generally makes use of heating an apparatus that is intended to distribute the heat to the product until it reaches the point of vaporization and can be inhaled.
Glass-Pipe Vaporizers
Glass-pipe vaporizers are glass pieces that are meant to be heated directly in order to indirectly disperse that heat into the product. The glass used must be thin enough for the heat to pass through its structure and potentially distribute the heat evenly. The piece must have a chamber within which the product will undergo vaporization. It must be assembled in a manner that will allow for air intake and output: The output being the inhalation nozzle, and the input being a sort of carb.
| the Improvisation of a Glass Pipe Vaporizer | |
|---|---|
"The Machine"
"The Machine" is essentially a glass vaporizer in which heat is meant to distribute through the pipe rather than across the glass. It utilizes a metal mesh plug inside of the pipe, on which the product is to be placed, melted and vaporized. The mesh acts both as a screen and a heat-sink, simultaneously allowing for the even heating of the product and prevention from inhaling unvaporized particulates.
This method of vaporization includes the standard variations—which are essentially the simple combination of a vaporizing bowl and a vapor chamber with an inhalation nozzle—and the bubbler variation in which the vapor passes through a water heat-sink before reaching the vapor chamber and inhalation nozzle.
Material Considerations:
- Copper will gradually turn black due to the formation of Copper(II) oxide (CuO). This reaction and its reactants are not detrimental to the process of administration.
Effective Use and Maintenance
As the term indicates, vaporizers are intended to vaporize, not to burn product, as such, the product should never come into contact with the flame or be overly heated to the point of burning. Generally, a vacuum must be generated in order to direct the heat through the product and to direct the vapor into the chamber. Which should not be allowed to sit for too long, as it may begin to precipitate within the chamber. Every toke should be held in the lungs as long as necessary for the vapor to be completely absorbed, as no vapor should be exhaled.
Heat-Source:
- Though many prefer a butane torch for expedient heating, and though many manners of vaporizer demand its use, a standard lighter will produce adequate heat for vaporization. A standard lighter is generally ideal for vaporizers that generate a strong enough vacuum to pull the flame toward the heat sink.
Common Methods of Inhalation:
- Some prefer to vaporize their entire dose before inhaling so as to administer one strong toke. This method can be quite harsh and difficult on the lungs and throat and may induce coughing. However, this is reportedly the most intense method of administration, almost always inducing a "breakthrough" if held in the lungs for adequate absorption.
- Others prefer to administer one dose within multiple tokes, which is reportedly slightly longer-lasting and much easier on the lungs and throat though possibly less intense. Usually the amount of tokes taken depends on the intended depth of the experience. In many cases, three is found to be more than adequate and four is said to be a "breakthrough" dose. Any subsequent tokes to the first toke must occur within mere minutes, as tolerance builds rapidly.
Cleaning the Apparatus:
- The apparatus used for vaporization can be cleaned using the same solvents used for extraction. However, volatility and toxicity concerns should be a strong consideration in choosing the proper solvent.
- It is preferable to choose a solvent that evaporates quickly and cleanly and able to dissolve a broad range of products, as oxidization is likely to have occurred.
- Residual solvent residue may be hazardous, as the user may inhale harmful fumes or potentially ignite the solvent.
- The residual product dissolved by the cleaning solvent may be salvaged by appropriate methods of crystallization. However, it is likely to contain a variety of inactive constituents, so proper purification measures would be advisable.
Concerns Regarding the Experience
- The primary effects of smoked DMT, induced within minutes of administration, last for about 5-10 minutes and are characterized by strong visual hallucination, a strong psychedelic quality, and minor auditory hallucination, while the secondary effects of a much more mild character may last for up to an hour.
- Tolerance is reportedly induced rapidly and often dissipates rapidly, though repeated or high doses may induce a slightly longer term tolerance.
- The experience of smoking DMT is said to be quite overwhelming. It is often necessary for the user to have a "sitter" nearby to tend to the smoking apparatus so as to not risk the possibility of the user damaging it or accidentally inflicting burns. It can also be temporarily debilitating, so the user may be advised to refrain from any physical activity.
- Set and setting are of the utmost importance, as the user is often rendered temporarily vulnerable and emotionally fragile. The user must determine the appropriate conditions in which administration is to take place but is advised to initially seek out a setting containing minimal stimuli.
- It may also behoove the user to seek out a more experienced sitter for guidance and support. Often this is unavailable, and as such, it would be more advisable for the user to perform the rite in solitude. An inexperienced sitter may inadvertently induce anxiety in the user out of negligence or unnecessary concern.
Further Elaboration and Technical Support
Potentiation
- The effects of DMT, as with most psychedelic substances, can be lengthened, strengthened, altered, or otherwise potentiated through the use of various psychoactive or bioactive compounds. The potentiation of DMT is actually the oldest method of administration, as its necessarily potentiated oral administration serves as the compound's longest known history of use. DMT is generally considered to be orally inactive without some form of potentiation, such as a harmaloid preparation. DMT's potentiation is not limited to oral use, however, as many of the same potentiating agents may be used in conjunction with vaporized or otherwise administered DMT.
Discussion of Harmaloids
Oral Administration
Discussion of "Jungle-Spice"
See also:
Further Potentiation
Appendices
Storage Concerns
DMT should be stored in a cool and dry place, preferably in a sealed container.
Discussion of Oxidization
Overview
Conversion
Administration of DMT N-Oxide
Analogues
5-MEO-DMT
Bufotenine
References
- ↑ L. M. Batista; R. N. Almeida; E. V. L. da-Cunha; M. S. da-Silva; J. M. Barbosa-Filho. Isolation and Identification of Putative Hallucinogenic Constituents from the Roots of Mimosa ophthalmocentra. In: Pharmaceutical Biology, Volume 37, Issue 1 January 1999