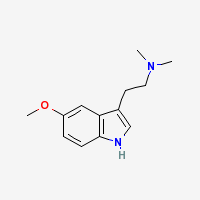
5-MeO-DMT
| Note: | This page has been transcluded to The Nexian DMT Handbook under the 5-MeO-DMT section or other locations within or without the handbook. Please markup in consideration of this. The top section header is to remain in place as a reference for subsequent section headers and to allow easy editing directly from the handbook. |
Contents
- 1 Brief overview - What is 5-MeO-DMT?
- 2 Chemical and physical properties
- 3 Effects
- 4 Pharmacology, toxicity and general safety
- 5 Life-forms containing 5-MeO-DMT
- 5.1 Acacia spp.
- 5.2 Anadenanthera spp.
- 5.3 Arundo spp.
- 5.4 Bromus spp.
- 5.5 Caesalpinia spp.
- 5.6 Delosperma spp.
- 5.7 Desmodium spp.
- 5.8 Dictyoloma spp.
- 5.9 Dutaillyea spp.
- 5.10 Digitaria spp.
- 5.11 Diplopterys spp.
- 5.12 Evodia spp.
- 5.13 Horsfieldiana spp.
- 5.14 Hugonia spp.
- 5.15 Iryanthera spp.
- 5.16 Justicia spp.
- 5.17 Lespedeza spp.
- 5.18 Mimosa spp.
- 5.19 Mucuna spp.
- 5.20 Osteophloem spp.
- 5.21 Other Grasses
- 5.22 Phalaris spp.
- 5.23 Sorghum spp.
- 5.24 Umbellularia spp.
- 5.25 Virola spp.
- 5.26 Humans and other animals
- 6 Extraction teks
- 7 Dosages and consumption methods
- 8 History of usage
- 9 Analysis of 5-MeO-DMT
- 10 Scientific publications
- 11 Other links of interest
Brief overview - What is 5-MeO-DMT?
5-MeO-DMT is a potent naturally occuring psychedelic alkaloid
Chemical and physical properties
For solubility, melting point, etc, check the 5-MeO-DMT Chemical and Physical Properties WIKI
5-MeO-DMT can be oxidized naturally or by the use of hydrogen peroxide and becomes 5-MeO-DMT N-Oxide
Effects
Excessively fast onset - 10-30 seconds.
Lower doses (2-6 mgs smoked) - Crushing body load quickly yielding a sense of body-orgasm. No CEV's, slight fractal visual distortions with open eyes. Peak effects 5-25 mins. Total time to full baseline 1.5-2.0 hours
Medium doses (7-15 mgs smoked) - Crushing body load yielding gasping and gagging nausea with any resistance and a sort of release into the 5-MeO state with surrender - beyond body-orgasm into gasping soul orgasm. CEV's show a sort of yellow or white field. Eyes want to open and close. Open eye visuals are best with living, loved ones. Fractalized in space and time. Peak effects 20-40 mins. Total time to full baseline 1.5-2.0 hours.
Higher doses (16-22 mgs smoked) - Body load that hits like a wall of drowning whiteness. Overwhelming. No time to resist or submit or feel nauseaus or groan or . . . Blackout for peak memories. Weird verbal outbursts or babbling. Extended peak effects, 20 mins to over an hour. Total time to baseline 2 hours to . . . . extended feeling of not being fully back or integrated that can last days, weeks or months.
It is worth noting that different anecdotal reports of 5-MeO-DMT use differ significantly in terms of their descriptions of the effects that particular dosages bring on. While some people have reported that 30 mgs produces only threshold effects, others have reported being completely overtaken by as little as 5 mgs. These drastic variations could be due to physical differences between individuals or variations in substance purity.
In order to maximize safety, it is recommended that potential users of 5-MeO-DMT start dosing themselves at the low end of the spectrum (2-6 mgs) before attempting higher dosages. As discussed earlier, even low doses of 5-MeO-DMT can occasionally produce extremely overwhelming effects.
Pharmacology, toxicity and general safety
Full agonist at 5-HT 1A, 5-HT 1c, 5-HT 1d & 5-HT 2, Callaway & McKenna 1998 5-HT receptor interactions & specificities: McKenna et at. 1990 Sec also: Glennon et al. 1979 & 1994; Sanders & Bush 1967
Distribution, mctabolism & excretion (in rat): Sanders & Bush 1967 Preferentially metabolized by MAO-A:Squires 1975; Suzuki et al. 1981
When ingested together with a MAOI, 5-MeO-DMT is O-demethylated by CYP2D6 and becomes Bufotenine in the liver, but without a MAOI, it is mostly N-Demethylated by MAOI into 5-MeO-Indole-acetic-acid (Shen et al 2010; Ai-Ming 2008).
There is some concern regarding taking 5-MeO-DMT orally, specially with a MAOI. There has been at least one reported death (Sklerov, 2005; Callaway 2006), and some hospitalizations. This might be due to individual metabolic differences. Check this thread for more info. If you are consuming 5-MeO-DMT orally for the first time, specially with MAOIs, start VERY low and raise your dosage gradually.
General For info on DMT safety, please reffer to Health and Safety tips regarding set and setting, how to integrate and so on, as written for DMT, are also relevant to 5-MeO-DMT
Life-forms containing 5-MeO-DMT
Acacia spp.
The alkaloid information about Acacia coming from TLC analysis is not to be considered definite. They are tentative identifications that need to be confirmed with GC-MS or other appropriate analytical methods. (ref Trout's Notes)
- Traces 5-MeO-DMT tentatively observed in twigs. 5 Oct. 1995. TLC by J. Appleseed 1995 (Xanthydrol), co-occuring with suspected DMT. (Trout's Notes)
Acacia angustissima Trace amounts tentatively observed in roots (unconfirmed) in March 1995. TLC by J. Appleseed. Not ob- served in second assay. Trace amounts in seeds. tlc by Appleseed 1995 (all with Xanthydrol)
Acacia auriculiformis Trace amounts tentative ly ()bserved in stem-bark (25 April 1995). tic by J. Appleseed 1995 (A band at this Rf was also seen in roots [mislabeled as Guaiacuml in 2 Sept. 1994 assay but used Ehrlichs reagent which does not differentiate between DMT and 5-MeO-DMT.)
Acacia cultriformis Blue band co-chromatographing with 5-MeO-DMT (Xanthydrol) Seen in branch stems, and in phyllodes, also in flowering spikes. Commercial florist's material. Sept 1996.
Blue band co-chromatograpbing with 5-MeO-DMT (Xanthydrol) Roots of two year old seedlings. TLC by J. Appleseed 1996. Also in stems Sept 1996. Not observed in roots Sept. 1996.
Acacia farnesiana Traces tentatively observed in green fruit. Not present in ripe fruit. (Xanthydrol) Also contained a suspected beta-carboline. (blue under UV) 24 July 1995. tlc by J. Appleseed 1995.
Acacia maidenii Traces observed in wood (October 1995); Observed in twigs (26 July in phyllode and in mixed phyllode and twigs 27 Oct 1995) tic by J. Appleseed 1995 (Xanthydrol) Co-occurring with suspected DMT in all samples of phyllodes and twigs but sole alkaloid seen when just using twigs. Not observed in bark or root bark. Note: Acacias such as this one do not produce leaves except when young or stressed. What are thought of as being leaves are actually phyllodes; a specially modified petiole (the short stalk at the leaf base.
Acacia nilorica
Faint traces observed in stem, roots and leaves. As-
sayed separately (Sept 1996; Xanthydrol)
Acacia obtusifolia
In some samp les as a minor component. Apparently
unpublished work. Pers. comm. with Snu Voogelbreinder. Needs confimation.
Appeared present in one sample of leaves / twigs looked
at by Mulga: Not observed in bark or root bark.
Acacia victoriae Strong blue band eo-chromatographing with 5-McO- DMT (Xanthydrol) Roots of two year old seedlings. tic by J. Appleseed 1996.
Anadenanthera spp.
Anadenanthera spp. (G. Seitz, 1965]
- Seedlings 0.001% 5-MeO-DMT [4% of 29 mg of total alkaloic / 100 gm dry.) (Schultes et al 1977 ref Trout's Notes)
- (As Piptadenia sp.) Caspar, 1964; Guapore, Brazil; Tupari. - Seeds: 0.11% 5-MeO-DMT (85% of 13 mg of total alkaloids / 100 grm) (Schultes et al 1977 ref Trout's Notes)
- 5-MeO-DMT reported in snuff thought to originate from this species. Conflicting reports. Most accounts have found only bufotenine in the seeds but several reports exist claiming the presence of DMT and/or 5-MeO-DMT. Torres et al. 1991 reported the detection of all three in snuff powder recovered from archaeological sites in Argentina believed to have been derived from A. colubrina seeds. Both A. colubrina and A. colubrina var. cebil occur in Argentina. While it is not clear which Torres and coworkers referred to; the latter is implied. No analysis of seeds or verifiable plant materials were reported in Torres et al. 1991.
- No.24625; Origin: Boa Vista, Brazil - Bark- 0.025% (25mg of 5-MeO-DMT / 100 gm. of dry bark]
Leaf- 0.006% [6 mg. of 5-MeO-DMT / 100 gm. of dry leaves] (Agurell et al. 1969 ref Trout's Notes)
- (as Piptadenia peregrina) Legler & Tschesche 1963 examined bark and reported 5-MeO-DMT formed 32% of the crude base. A mixture of 5-MeO-NMT and NMT comprised 41%. Used Brazilian material. Seeds collected in western Brazil in the Rio Branco region during 1953 5-MeO-DMT ; Bark collected in Colombia during 1956 with 5-MeO-DMT (Holmstedt & Lindgren 1967 ref Trout's Notes)
- No number; La Carolina, Barrio St. Just, near San Juan, Puerto Rico, December 1974. Same colony as Schultes 26363. - Mature seeds collected in March 1975; hill behind El Comandan te horse-racing track. 1975 analysis (5 months after collection): 5-MeO-DMT- 1% of total alkaloid. 1977 analysis of same material could detect only bufotenine (80% of total alkaloid in 1975). (Schultes et al 1977 ref Trout's Notes)
- R. E. Schultes 26363; La Carolina, Barrio St. Just, near San Juan, Puerto Rico, Dec. 1972 - Immature seeds collected December 1972 0.04% 5-MeO-DMT (19% of 209 mg of total alkaloid/ 100 gm dry)
Seedlings 0.024% 5-MeO-DMT (95% of25 mg of total alkaloids / 100 gm dry] Pods without seeds 0.012% 5-MeO-DMT (91% of 13 mg of total alkaloids / 100 gm dry] Leaves 0.094% 5-MeO-DMT (88% of 107mg oftotal alkaloids / 100 gm dry] Twigs 0.0357% 5-MeO-DMT (94% of38 mg of total alkaloids / 100 gm dry) Bark (0.41% total alkaloid) 0.39% 5-MeO-DMT (95% of 410 mg of total alkaloids / 100 gm dry) Roots (0.69% total alkaloid) 0.678% 5-MeO-DMT (97% of 699 mg of total alkaloid / 100 gm dry] (Schultes et al. 1977 ref Trout's notes)
- R.E.Schultes 24625; Boa Vista, Brazil)
Leaves 0.00624% 5-MeO-DMT (48% of 13 mg of total alkaloids / 100 gm dry] Bark 0.025% 5-MeO-DMT (59% of 42 mg of total alkaloids / 100 gm dry) (Schultes et al. 1977 ref Trout's notes)
- Snuff: "epena"
Obtained from the Waica by George Seitz. 5-MeO-DMT was the major alkaloid. Bufotenine was present as a minor component. Because of this, the claimed plant source (Virola) has been questioned. Holmstedt 1965 Many snuff using people use both snuffs, although many have preferences one way or the other. While idle speculation; we wonder if preparation might not have involved both plants, or if the snuff witnessed as made from one source ( Virola) may not have been contaminated with residuals of ano ther snuff (Anadenanthera) during processing or storage, in- volved the admixture of other Myristicaeaous plants or as-yet unidentified plants, or perhaps have been derived from an ·altogether different but as yet un- known source.
- Snuff: "parica"
Snuff as prepared by Piaroa Indians (collected 1955) 5-MeO-DMT [with DMT and Bufotenine] Holmstedt & Lindgren 1967
- Snuff: "yopo"
Snuff prepared, by the Pix.asi-teri (or Bisashi-teri) of Upper Orinoco, from the seeds of an Anaden.anthera species. 5-MeO-DMT [with Bufotenine) Marini-Bettolo eta/. 1964
- Snuff: "yopo"
Snuff collected in Colombia (collected 1956) 5-MeO-DMT [with DMT and Bufotenine] Holmstedt & Lindgren 1967
Arundo spp.
Arundo donax Leaf and flower. (Ghosal 1972a ref Trout's Notes)
Bromus spp.
(Brome grass)
5-MeO-DMT appeared potentially present in at least some specimens of one Bromus sp. (B. breviaristatus) 1996 tlc by J. Appleseed. Species was grown by Giorgio Samorini from seeds provided by Trout and identified at seed maturity by Dr. F. Festi in 1999. (Ref Trout's Notes)
Caesalpinia spp.
- In stem-bark (with 1 other band) 1996 assays.
- In roots. (Nice band with two others present) June
1995, also 1996. tlc by J. Appleseed (Xanthydrol) (Ref. Trout's Notes)
- In flowers and buds. (26 August 1995) tlc by J.
Appleseed 1995. [August 1994 of a small sample of dead flower petals did not detect this alkaloid but did show a faint indolic band at a lower Rf.] In roots (nice band) June 1995, also 1996. tlc by J. Appleseed 1995 (Xanthydrol) Desmodium sp. (Wild species, not yet positively iden- tified; Austin, Tx.) Trace amounts in aerial portions (co-occurring with suspected DMT). tlc with Xanthydro1 spray, 24 June 1995. (Ref. Trout's Notes)
Delosperma spp.
TLC assays by Johnny Appleseed (ref Trout's Notes)
- May 1995 assay, 5-MeO-DMT Faint (Xanthydrol) Nov. 1995 assay: Dark blue and purple band corresponding to DMT and 5-MeO-DMT. (Xanthydrol) Other earlier positives used Ehrlich's.
- 2 Nov. 1995 sample. 5-MeO-DMT, Very nice dark band with Xanthydrol.
Delosperma cooperi May 1995 assay (two sources) also in 2 Nov. 1995 sample. 3 positives total with Xanthydrol. Other positives using Ehrlichs; co-occurrence with DMT seen in 2 Nov. 1995 sample.) (Sasha did not confirm on material from another source.
- 5-MeO-DMT Nice single blue band. Xanthydrol. Sept. 1996.
- 5-MeO-DMT. 2 Nov. 1995 sample. Dark band with Xanthydrol.
Delosperma harazianun Audhali Plateau, Yemen
- 5-MeO-DMT. 2 Nov. 1995 sampling. Dark band with Xanthydrol.(Co-occurring with DMT) Not observed In D. harazianum Shibam.)
- Nice 5-MeO-DMT band.· Xanthydrol. Sept. 1996. (Co-occurrence with DMT)
- 2 Nov. 1995 Sample. 5-MeO-DMT. Dark band with Xanthydrol.
- 9 May 1995. Weak 5-MeO band. (Xanthydrol)
- (Same individual plant tested Dec. 1994)
Positives: May and 2 Nov. 1995. [Faint in Nov. Good in May] (Xanthydrol.). Material harvested Aug. and Dec. 1995 tested positive (with DMT co-occurrence); Sept. 96 (also using Xanthydrol). Assay with Ehrlich's had shown decent band at this Rf in 5 Dec. 1994 sampling.
Desmodium spp.
- Aerial parts (0.057% by wet weight; 0.57 gm. from 1kg. of fresh wet material.) Banerjee & Ghosal 1969 (Ref. Trout's Notes)
Stem/leaf (Ghosal 1972a Ref. Trout's Notes)
- Green Plant (Stem and Leaf) Ghosal & Bhattachaya 1972 (Ref. Trout's Notes)
- Our assays of seedgrown plants detected 5-MeO-DMT as present in trace amounts in seeds
(May 1995 assay). No alkaloids were observed iu this species until after it was 2 years old. 5-MeO-DMT was observed in small amounts in roots and also in stems (May 1995); Also in leaf Feb. 1995 (using large red leaves left from winter) and May 1995 using normally colored new but full sized leaves. Leaves also tested positive (faint) in Nov. 1995. tlc by J. Appleseed using Xanthydrol. (Ref. Trout's Notes)
- Leaves (35+ mg. from 2 kg) Ghosal e1 al 1972a (Ref. Trout's Notes)
ln stem/leaves Ghosal 1972a (Ref. Trout's Notes) Roots Gihosal 1972a (Ref. Trout's Notes)
Desmodium pulchellum Bentham ex Baker
- Whole plant (0.2-0.25% by dry weight.) Ghosal &
Mukherjee 1964 (Major alkaloid. Ghosal & Mukherjee 1965. (Amount not given. Plates crystallized from 8.36 grams of impure chromatographic fraction resi- due; from 4 kg of dried whole plant.) Ghosal & Mukherjee 1966 (Ref. Trout's Notes) Stem and leaf of young seedl ing [Trace.] Ghosal et at. 1972c Stem and leaf of mature plant [0.476% by dry weight; 34% of 1.4% Total alkaloid] Ghosal eta!. 1972c Root of mature plant [0.132% by dry weight; J 2% of 1.1% Total alkaloid] (Also, in same paper; 1.8 kg. of dried roots yielded 0.23 gm; i.e. - 0.013% by dry weight.) Ghosal et a/. 1972c Seeds (ripe) of mature plant [0.002% by dry weight; 10% of0.02% Total alkaloidJ Ghosal eta/. 1972c Root, stem-leaf and flower (A mounts not given) Ghosal 1972a
- Whole plant. (Amount not given) Hsli et a/. 1982
cited HsU 1970 [Source article has not been located. Title is suspect.]
Dictyoloma spp.
Dictyoloma incanescens· DC 0.04% isolated from dry bark collected in winter near Rio de Janeiro. Voucher prepared. (Pachter et al 1959 ref Trout's Notes)
Dutaillyea spp.
Dutaillyea drupacea (Baillon) Hartley New Caledonia
- 5-MeO-DMT was the sole alkaloid (98%) in the leaves- 0.04%. [The published value of 0.4%, repeatedly appearing in the literature, IS A TYPO] (Baudouin et al. 1981 ref Trout's Notes)
- 5-MeO-DMT was absent from trunk-bark (Baudouin et al. 1981 ref Trout's Notes)
Dutaillyea oreophila (Baillon) Sevenet-Pusset New Caledonia
- Leaves- 0.02% 5-MeO-DMT by dry weight. Major alkaloid (40% of 0.05% total alkaloids) Co-occurring with 2-Methyl-6-methoxy-tetrahydro-beta-carboline (0.005% dry wt), Hordenine (0.0125% dry wt.) and Kokusagine (0.0125% dry wt.) (Baudouin et al. 1981 ref Trout's Notes)
- 5-MeO-DMT was absent from trunk-bark (Baudouin et al. 1981 ref Trout's Notes)
Digitaria spp.
(Crab grass)
Digitaria sanguinalis 5-MeO-DMT appeared potentially present in at least one local species; probably D. sanguinalis. Positive identification pending. 1996 tlc by J. Appleseed. (Ref Trout's Notes)
Diplopterys spp.
- Not found by Mckenna 1984, who only found DMT and traces of bufotenine
- Not found by Endlessness who only found major DMT peak (small peaks in process of identification but are NOT 5-MeO-DMT).
- 0.4655% DMT, co-occuring with traces of NMT, Bufotenine and MTHBC (Agurell et al. 1968) ( misidentified as Banisteriopsis rusbyana see Gates 1982)
- 0.16638% DMT, 0.0035% MTHBC and 0.0035% 5-MeO-DMT in dried stem (Agurell et al. 1968) ( misidentified as Banisteriopsis rusbyana see Gates 1982)
Evodia spp.
- 5-MeO-DMT in unripe fruit. Takagi et al. 1979 recovered 30 mg from 10 kg unripe fruit. Yu et al. 1997 reported 0.00015% by dry weight in the unripe fruit [ 15 mg from 3 kg.l
- Also present in the roots. Shulgin & Shulgin 1997
- 5-MeO-DMT (0.21% dry wt.) is the major alkaloid (35% of 0.61% total alkaloids) in aerial parts. Accom panied by 0.03% 5-MeO-DMT-N-oxide and 0.024% 2-Methyl-6-methoxy-tetrahydro-beta-carboline, Acronydine (0.61%), Kokusaginine (0.183%), Acronycidine (0.0 12%), Melicopicine (0.043%), Melicopidine, 0.018%) and Acronycine (0.006%). (Skaltsounis et al. 1983 ref Trout's Notes)
Horsfieldiana spp.
Horsfieldiana superba (Hk. f. ctTh.) Warb.
- Collected in September at Sandakan, Sabah, Malaysia.)
5-MeO-DMT as minor leaf alkaloid (0.0007%; 20mg from 2.8 kg of lea ves.] Roots and bark apparently unexamined. (Jossang et al 1991 ref Trout's Notes)
Hugonia spp.
Hugonia oreogena Schleeter
- Traces of 5-MeO-DMT in rootbark. (0.5 mg from 140 grams) (Ikhiri et al. 1987 ref Trout's Notes)
Iryanthera spp.
- Trace amounts in bark Holmstedt et al. 1980
Justicia spp.
- In leaf. Shulgin & Shulgin 1997
- var. stenophylla -
Leaf (dry) sampled 2 Nov. 1995 showed a faint blue band that co-chromatographed with 5-MeO-DMT. Plant from LER. TLC by Applesecd 1995. (Xanthydrol) We wonder if the above might not actually be the same alkaloid as McKenna observed rather than 5-MeO-DMT. While TLC has indicated DMT traces in this SAME plant, this was our only sampling to show this compound and no DMT.
Lespedeza spp.
Lespedeza bicolor Turcaninow var japonica Nakai
- In leaf and root bark. Smith cited Goto et al 1958 (Ref Trout's Notes)
- Present in root bark (Less than 0.1%). Morimoto & Matsumoto 1966 (Ref Trout's Notes)
Lespedeza bicolor Our assays did not detect any alkaloids in any parts during the first year. ( 1994) We did detect 5-MeO-DMT in both seeds and mixed seeds and pods. (May 1995) tlc by J Appleseed. See comments under DMT as we may have mistaken the two as tlc used Eh.rlichs (Ref Trout's Notes)
Mimosa spp.
- 5-MeO-DMT in low amounts in stem and leaf after the first year
(November harvest of 15 month old plants). Concentrations were higher in the roots (August harvest). Assays 2 Nov. 1995 Very young seedlings (whole plant) tested in 1996 showed a very dark suspected 5-MeO-DMT band.
- In branches; 1996 assays.
tlc by J. Appleseed 1995-6 (ref trout's Notes) [DMT did not start to show up in assays until after second year, at which time it was present in leaf and root.]
- Maybe appearing in error? Not observed by Gupta and coworkers
Mimosa tenuiflora (Willd.) Poir. The listing of 5-MeO-DMT is in error. Meckes-Lozoya ran it as a pure reference sample only.
Mucuna spp.
- 0.01% in fresh leaves Ghosal et al 1971
- In leaf seed, stem and roots. Ott cited Bhattacharya et al 1971; Ghosal 1972a; Ghosal et al. 1971
- In root, stem-leaf and pod Ghosal 1972a
Osteophloem spp.
Osteophloem platyspermum (DC) Warb. (Schultes &Rodrigues #26126]
- One of 3 alkaloids in 0.62 mg of total alkaloid from 100 grams of dry bark. (Brazil);
- Other material: [Schultes & Tovar #7095 (Peru)] tested negative with Ehrlich's and Dragendorff's. (Holmstedt et al. 1980)
- McKenna et al 1984b tested a samples of leaves DMK-78 but failed to find. Did report Methyltryptophan methyl ester
Other Grasses
??? spp. (Wild Rye, Winter Rye, Rye Grass) 5-MeO-DMT appeared to be potentially present in several local species (2-4 spp.; including both annual & perennial ryes. Including the haying material called "coastal') 1996 tlc by J. Appleseed. (Ref Trout's Notes)
Phalaris spp.
Phalaris data below is incomplete. Alkaloid concentrations and proportions are highly variable from year to year and show dramatic seasonal fluctuations. Concentrations between plant parts and first growth versus regrowih are also very different. In many populations there may be marked differences in both the amounts present and in actual alkaloid profile from one plant to the next. (i.e. plants in the same population and arising from the same seeds may show completely different chemistry, not simply differing concentrations.) See the amazing Festi & Samorini pieces listed in the Phalaris spp. page about cultivation for a review and overview of what is known so far.
- A major alkaloid in all samples they examined. (Culvenor et al. 1964)
- 5-MeO-DMT was not present in all clones examined (4 out of 12) Frahn & Illman 1973
- 5-MeO-DMT in leaf. 0.01-0.28% in material from California. (Festi & Samorini 1994a cited Welch 1971 ref Trout's Notes)
- var. AQ-1 (Italy) Weak occurrence reported (HPLC). (Festi & Samorini 1994b ref Trout's Notes)
- as Phalaris tuberosa - Strong in leaf. 2 Nov. and 17 Sept. 1995. Dec 1995. Assay. tlc by J. Appleseed 1995 (ref Trout's Notes)
- var Killer Phalaris (At one point this was synonymous with cv. Uneta, but has been in uncontrolled propagation for long enough this is no longer certain.) 5-MeO-DMT predominated in 25 June, 17 Sept., 2 Nov. 1995 samples. (DMT was predominate alkaloid in Fall 1994 by TLC) (ref Trout's Notes)
- Clone #R5 "large" amount of DMT (5-MeO? possible mistake in Trout's Notes) co-occurring with twice amount of 2-MTHBC (Clone, designated 405-9, originating with U.S. Regional Pasture Research Laboratory, University Park, Pennsylvania)
- Clone #R37 ·'trace" amount of 5-MeO-DMT co-occurring with " intermediate" amounts of hordenine and "large" amounts of 6-methoxy-2,9-dimethyl-1,2,3,4-
tetrahydro-beta-carboline. (From highly diverse source population used in plant breeding and genetic studies at the University of Minnesota. Department of Agronomy and Plant Generics".)
- Clone #R51 ·'large" amount of 5-MeO-DMT as sole observed al.kaloid. [Same source as R37)
- Clone #R96 " large" amount of 5-MeO-DMT co-occurring with "trace" amounts of hordenine and "trace" amounts of 6-methoxy-2,9-dimethyl-1,2,3,4-tetrahydro-beta-carboline. (Same source as R37) (Gander et al. 1976. ref Trout's Notes)
- cv. Australian Commercial (CPI 119305) A major alkaloid in 7 day old seedlings. 150 nmol / 100 seedlings. (Mulvena & Slaytor 1983 ref Trout's Notes); In seedlings. (Mack et al. 1988); Mature 0.05% dry wt.(Baxter & Slaytor 1972)
- cv. Sirocco 51 nmol / 100 seedlings. (Mulvena & Slaytor 1983 ref Trout's Notes); Major base. (Frahn & O'Keefe 1971); 5-MeO-DMT in leaf (Ott 1994 cited Culvenor et al 1964; Baxter & Slaytor 1972; frahn & Illman 1973 ; Moore et al 1967; Mulvena & Slaytor 1982; Oram & Williams 1967 ref Trout's Notes)
- 5-MeO-DMT in leaf and whole plant Barnes et al 1971; Culvenor et al 1964; Gander et al. 1976; Majak & Bose 1977; Majak et a!. 1978; Marten et al. 1973
Williams et al. 197 1. Many others.
- 0.0002-0.0067% 5-MeO-DMT in material from British Columbia. (Majak & Bose 1977)
- 0-0.02% in material from Minnesota. NRG741 was strongest of those tested & NRG721 the weakest. (Majak eta/. 1978)
Festi & Samorini 1994a
- tlc by Johnny Appleseed
- P.I. 172442 Turkey (cv. Turkey Red)0.0025% to 0.045% total alkaloid by wet weight. 5-MeO is predominate alkaloid. (J.Appleseed (undated manuscript); "Ayahuasca analog plants of the temperate zone." Tlc by Johnny Appleseed: fall 1994, 25 June, 17 Sept, 2 Nov. 1995. (ref Trout's Notes)
- cv Ottawa Synthetic : 5-MeO-DMT present, Amounts not given. Detected by TLC (Woods & Clark 1971 ref Trout's Notes)
- PI 202676, PI 231044: 5-MeO-DMT reported, Appleseed tlc evaluation of field trials using USDA seeds. (ref Trout's Notes)
- PI 167261, and also 284185 (lower levels) 5-MeO-DMT reported, Appleseed tlc evaluation of field trials using USDA seeds. (ref Trout's Notes)
- Traces reported (HPLC). Festi & Samorini 1994b
Phalaris stenoptera (= P tuberosa var. stenoptera)
- Variable amounts. Festi & Samorini 1994b cited Rendig et al 1970 as finding 135-264 J.Lg/ml of expressed juice. (ref Trout's Notes)
Sorghum spp.
Sorghum halepense (Johnson Grass, Aleppo Grass, Egyptian Millet, Grass Sorghum, Means Grass)
- Low levels of 5-Me0-DMT in leaf only. 1996 TLC by J. Appleseed. (Xanthydrol) Samples collected spring and summer; Central Texas. (ref Trout's Notes)
Umbellularia spp.
Umbellularia californica (Hook & Am.) Nutt.
- Unspecified concentration of 5-MeO-DMT in stembark. Needs confirmation. (Ratsch 1998 ref Trout's Notes)
Virola spp.
Snuff and other products from unidentified Virola.
- Virola based arrow poison (Yanomamo). 8% 5-MeO-DMT by dry wt. (Macrae & Towers 1984 cited Galeffi et al 1983 ref Trout's Notes)
- "epena" - Snuff as prepared by Araraibo Indians (collected 1965) - 5-MeO-DMT [with DMT]
- Snuff as prepared by Tucano Indians (collected 1965) - 5-MeO-DMT [with DMT and 5-MeO-MMT)
- Snuff as prepared by Waica Indians (collected 1965) - 5-MeO-DMT [with NMT and DMT] (Holmstedt & Lindgren 1967 ref Trout's Notes
- epena" [N.24574; Origin: Rio Cauaburi, Brazil) - 0.5% 5-MeO-DMT [515 mg. of 5-MeO-DMT / 100g snuff) (Agurell et al. 1969 ref Trout's Notes)
- nyakwana (n24626; Origin: Tototobi, Brazil] - 9.68% 5-MeO-DMT [9,6130 mg. of 5-MeO-DMT / 100g snuff) (Agurell et al. 1969 ref Trout's Notes)
- Suspected virola snuff (Based on chemistry, not voucher) - 1.4% 5-MeO-DMT. (De Budowski et al. 1974 ref Trout's Notes)
- Virola paste (No voucher; " oo'-koey" ; La Chorrera) - 5-MeO-DMT 1.19 mg/ ml. DMT also present at O.3 mg/ ml (Mckenna et al 1984a ref Trout's Notes)
- (Collected on March 12; Santa Rose, Iquitos, on Rio Momon in Peru) Wood was found to contain 5-MeO-DMT (major alkaloid) co-occurring with 2-Methvl-6-methoxy-1,2,3,4-tetrahydro-Beta-carboline and nicotine. No amounts given. Miles et al 1987 ref Trout's Notes
- Plowman, Schultes and Tovar #7093; Origin: Pebas, Peru.) (5-MeO ?) DMT in bark Holmstedt et al. 1980 ref Trout's Notes
- 5-MeO-DMT has NOT been found on all samples
- bark 0.245 mg of 5-Me0-DMT/ gm dry bark. Macrae & towers 1984 cited McKenna et al. 1984a
- Plowman, Schultes and Tovar #6920; Origin: Pebas, Peru. Present in resin and phloem (Not in leaves) Holmstedt et al. 1980 ref Trout's Notes
- Plowman, Schultes and Tovar #7263; Origin: Pebas, Peru. Present in paste and phloem. Holmstedt et al. 1980 ref Trout's Notes
- Plowman, Schultes and Tovar #7092; Origin: Pebas, Peru. Trace in bark. Holmstedt et al. 1980 ref Trout's Notes
- DMK-59. 5-MeO-DMT present in bark (not in leaves- DMT and NMT present in leaves). McKenna et al. 1984b
- DMK-67, DMK-68 and DMK-69. 5-MeO-DMT in leaf (Negative assay in bark) McKenna et al. 1984b
- Paste - DMK-59; Alfredo Moreno no. I. 5-MeO-DMT 2.03 mg/ ml (Sole base present). McKenna et al. 1984a ref Trout's Notes
- Many collections of this species were reported devoid of alkaloid. Others contained only traces of DMT.
- [No.24614; Origin : Manaus, Brazil) - Roots-0.00059% [0.59 mg. of 5-Me0-DMT/ 100 gr dry wt) Agurell et al 1969 ref Trout's Notes.
- (Schultes No. 24616; Origin Manaus, Brazil] - Trace amounts of 5-MeO-DMT in dry roots. 590ug per 100 grams. Holmstedt et al. 1980 ref Trout's Notes
- 5-MeO-DMT In bark and paste. Holmstedt et al. 1980 ref Trout's Notes
- 5-MeO-DMT in plant. Part and amount not given. In roots (even lower levels). Lai et al. 1973 ref Trout's Notes
- [Schultes N.24612; Origin: Manaus, Brazil). Bark- 0.008%? [8 mg. of 5-MeO-DMT per 100g bark) given in Holmstedt et al. (1980) as 190 mg per 100 grams of dry bark which, if true, would be 0.19%.)
- Roots- 0.135% [ 135 mg. of 5-MeO-DMT per 100g root) - Agurell et al 1969
- Sample DMK-40. Found 5-MeO-DMT in bark. McKenna et al. 1984b
- Schultes No. 24595; Origin: Manaus, Brazil : * Bark- 0.11% [108 mg. of 5-MeO-DMT/ 100 gm. ) * Roots- 0.011% [ 11 mg. of 5-methoxy-DMT/ 100 gm.]
- Leaves - Traces. Agurell et al. 1969
- Schultes No. 24626; Origin: Tototobi, Brazil] * Bark- 0.062% [62 mg. of 5-Me0-DMT / 100 gm.) Agurell et al. 1969 ref Trout's Notes. Ott 1993 cited Holmstedt 1965
- Schultes No. 24613; Origin: Manaus, Brazil. * Roots- 0.001% [1 mg per 100 gm.]* Leaves- <0.001% [Less than 1 mg per 100 grams. (Holmstedt at al. 1980 listed only DMT in leaves for this same collection)]
- No. 24613; Origin: Manaus, Brazil. * Roots- 0.001% [1 mg per 100 gm.]* Leaves- <0.001% (Less than 1 mg per 100 grams. (Holmstedt et a. 1980 listed only DMT in leaves for this same collection) Agurell et al. 1969 ref Trout's Notes (COPY OF ABOVE ANALYSIS FROM V. THEIODORA? MISTAKE?)
- Plowman, Schultes and Tovar N7094; Origin: Pebas, Peru): * Bark- 0.0042% [4.1mg per 100 grams of dry roots. * Leaf- 0.0003% [0.29 mg per 100 grams of dry leaves] Holmstedt et at. 1980 ref Trout's Notes
Humans and other animals
- Cutaneous glands were found to contain as much as 60 to
160 mg per gram of dry tissue 1.0-3.5 mg/ gm of non-glandular skin. (Erspamer et al. 1965 ref Trout's Notes)
- As much as 5- 15% of parotid and coxal glands by dry
weight. Ranged from 50-1 50 mg per gram oflarge cutaneous glands (dry) and from 0.42-3.5 mg per gram in the rest of the dry skin. (Erspamer et at. 1967 ref Trout's Notes (occurs with its N-sulfate)
- Sample analysis of human cerebrospinal fluid included 5-MeO-DMT. (Christian et al. 1975 ref Trout's Notes)
- Found in cerebrospinal fluid of some psychotics and a few normal people by Corbett et al. 1978. Narasimhachari et
al. 1971b reported it was more common in psychotics than normals (ref Trout's Notes)
- Also detected in some patients by Smythies et al. 1979 but it is not clear in their account exactly which subjects showed its presence and which did not. (ref Trout's Notes)
Extraction teks
Any typical extraction teks for DMT should potentially extract 5-MeO-DMT (but it will also extract DMT together). For separating 5-MeO-DMT from DMT, still no fail-proof method has been developed. Column chromatography will work using acetic acid as an eluent and silica as mobile phase. DMT will elute first (Rf 0.5), then 5-MeO-DMT (Rf 0.6) and bufotenine last (Rf 0.65) (De Bukowski 1974 ref Trout's Notes)
Dosages and consumption methods
5-MeO-DMT is around 5 times more potent than DMT by weight.
Smoked, dosage is around 5-15mg.
Oral, according to Jonathan Ott, 30mg are active without the need for MAOIs. Mixing with MAOIs, it will be 3 times stronger, so a dosage will be around 10mg. There is some concern about 5-MeO-DMT's oral ingestion safety, and it might be related with individual metabolism differences. Check this thread for more info. If you are consuming 5-MeO-DMT orally for the first time, specially with MAOIs, start VERY low and raise your dosage gradually.
Insufflated, 5-20mg (Erowid)
History of usage
Analysis of 5-MeO-DMT
Colorimetric
Reagent color results here
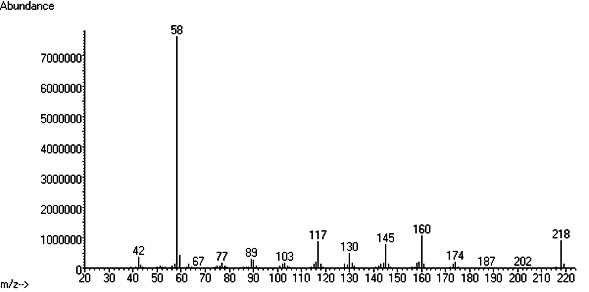
GC-MS
Retention time: 12.946 (System used)
Other info: 5-MeO-DMT: EI/MS (m/z, %): 218 (M+ , 10), 160 (6.3), 145 (2.5), 117 (5.0), 58 (100), 42 (4). (Takahashi 2008)
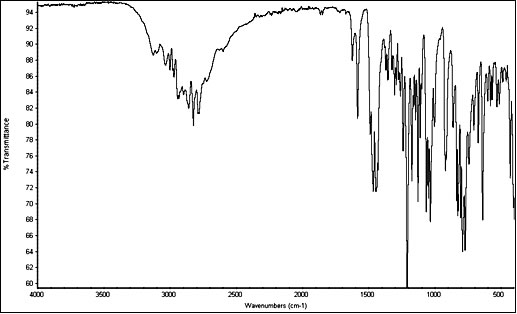
InfraRed
5-MeO-DMT freebase
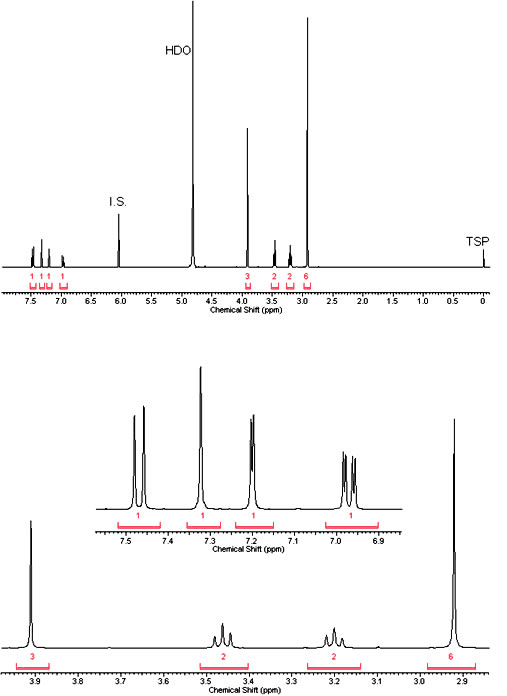
NMR
1H NMR (400 MHz, D2O) δ ppm 7.47 (d, J=8.90 Hz, 1 H) 7.32 (s, 1 H) 7.20 (d, J=2.45 Hz, 1 H) 6.97 (dd, J=8.90, 2.45 Hz, 1 H) 3.91 (s, 3 H) 3.46 (t, J=7.43 Hz, 2 H) 3.20 (t, J=7.43 Hz, 2 H) 2.92 (s, 6 H).
(Source: Microgram Bulletin Vol. 3, Pg 8)
Other info: 1 H-NMR (CDCl , δ): 2.34 (6 H, s), 2.63 (2 H, m), 2.91 (2 H, m), 3.86 (3 H, s), 6.85 (1 H, dd, J = 2.3, 8.6 Hz), 6.98 (1 H, br d, J = 2.3 Hz), 7.05 (1 H, d, J = 2.3 Hz), 7.22 (1 H, d, J = 8.6 Hz). 13 C-NMR (CDCl3 , δ): 23.7, 45.4, 56.0, 60.1, 100.8, 111.8, 112.1, 113.9, 122.3, 127.8, 131.5, 153.9. (Takahashi 2008)