Difference between revisions of "5-MeO-DMT"
Endlessness (Talk | contribs) (→Plants containing 5-MeO-DMT) |
Endlessness (Talk | contribs) (→Arundo spp.) |
||
| Line 107: | Line 107: | ||
Leaf and flower. (Ghosal 1972a ref Trout's Notes) | Leaf and flower. (Ghosal 1972a ref Trout's Notes) | ||
| − | === Bromus spp. === (Brome grass) | + | === Bromus spp. === (Brome grass) |
[[Bromus breviaristatus]] | [[Bromus breviaristatus]] | ||
| Line 114: | Line 114: | ||
| − | === Digitaria spp.=== (Crab grass) | + | === Digitaria spp.=== (Crab grass) |
[[Digitaria sanguinalis]] | [[Digitaria sanguinalis]] | ||
Revision as of 15:24, 13 November 2011
| Note: | This page has been transcluded to The Nexian DMT Handbook under the 5-MeO-DMT section or other locations within or without the handbook. Please markup in consideration of this. The top section header is to remain in place as a reference for subsequent section headers and to allow easy editing directly from the handbook. |
Contents
- 1 Brief overview - What is 5-MeO-DMT?
- 2 Chemical and physical properties
- 3 Effects
- 4 Pharmacology, toxicity and general safety
- 5 Plants containing 5-MeO-DMT
- 6 Extraction teks
- 7 Dosages and consumption methods
- 8 History of usage
- 9 Analysis of 5-MeO-DMT
- 10 Scientific publications
- 11 Other links of interest
Brief overview - What is 5-MeO-DMT?
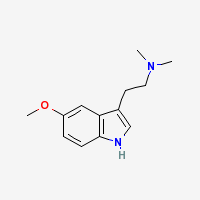
5-MeO-DMT is a potent naturally occuring psychedelic alkaloid
Chemical and physical properties
For solubility, melting point, etc, check the 5-MeO-DMT Chemical and Physical Properties WIKI
Effects
Excessively fast onset - 10-30 seconds.
Lower doses (2-6 mgs smoked) - Crushing body load quickly yielding a sense of body-orgasm. No CEV's, slight fractal visual distortions with open eyes. Peak effects 5-25 mins. Total time to full baseline 1.5-2.0 hours
Medium doses (7-15 mgs smoked) - Crushing body load yielding gasping and gagging nausea with any resistance and a sort of release into the 5-meo state with surrender - beyond body-orgasm into gasping soul orgasm. CEV's show a sort of yellow or white field. Eyes want to open and close. Open eye visuals are best with living, loved ones. Fractalized in space and time. Peak effects 20-40 mins. Total time to full baseline 1.5-2.0 hours.
Higher doses (16-22 mgs smoked) - Body load that hits like a wall of drowning whiteness. Overwhelming. No time to resist or submit or feel nauseaus or groan or . . . Blackout for peak memories. Weird verbal outbursts or babbling. Extended peak effects, 20 mins to over an hour. Total time to baseline 2 hours to . . . . extended feeling of not being fully back or integrated that can last days, weeks or months.
Pharmacology, toxicity and general safety
When ingested together with a MAOI, 5-MeO-DMT is O-demethylated by CYP2D6 and becomes Bufotenine in the liver, but without a MAOI, it is mostly N-Demethylated by MAOI into 5-MeO-Indole-acetic-acid (Shen et al 2010; Ai-Ming 2008).
There is some concern regarding taking 5-MeO-DMT orally, specially with a MAOI. There has been at least one reported death (Sklerov, 2005; Callaway 2006), and some hospitalizations. This might be due to individual metabolic differences. Check this thread for more info. If you are consuming 5-MeO-DMT orally for the first time, specially with MAOIs, start VERY low and raise your dosage gradually.
General Health and Safety tips regarding set and setting, how to integrate and so on, as written for DMT, are also relevant to 5-MeO-DMT
Plants containing 5-MeO-DMT
Acacia spp.
The alkaloid information about Acacia coming from TLC analysis is not to be considered definite. They are tentative identifications that need to be confirmed with GC-MS or other appropriate analytical methods.
- Traces 5-MeO-DMT tentatively observed in twigs. 5 Oct. 1995. TLC by J. Appleseed 1995 (Xanthydrol), co-occuring with suspected DMT. (Trout's Notes)
Acacia angustissima Trace amounts tentatively observed in roots (unconfirmed) in March 1995. TLC by J. Appleseed. Not ob- served in second assay. Trace amounts in seeds. tic by Appleseed 1995 (all with Xanthydrol)
Acacia auriculiformis Trace amounts tentative ly ()bserved in stem-bark (25 April 1995). tic by J. Appleseed 1995 (A band at this Rfwas a lso seen in roots [mislabeled as Guaiacuml in 2 Sept. 1994 assay but used Ehrlichs reagent which does not differentiate between DMT and 5-McO- DMT.)
Acacia cultriformis Blue band co-chromatographing with 5-MeO-DMT (Xanthydrol) Seen in branch stems, and in ph yllodes, also in flowering spikes. Commercial florist's material. Sept 1996.
Blue band co-chromatograpbing with 5-MeO-DMT (Xanthydrol) Roots of two year old seedlings. lie by J. Appleseed 1996. A Iso in stems Sept 1996. Not observed in roots Sept. 1996.
Acaciafarnesiana Traces tentatively observed in green fruit. Not present in ripe fruit. (Xanthydrol) Also contained a suspected f3-carboline. (blue under UV) 24 July 1995. tic by J. Appleseed 1995.
Acacia maidenii Traces observed in wood (October 1995); Observed in twigs (26 July in phyllode and in mixed phyllode and twigs 27 Oct 1995) tic by J. Applcseed 1995 (Xanthydrol) Co-occurring with suspected DMT in all samples of phyllodesltwig but sole alkaloid seen when just using twigs. Not observed in bark or root bark. [Note: Acacias such as this one do not produce leaves except when young or stressed. What are thought of as being leaves are actually phyllodes; a specially modified petiole (the short stalk at the leaf base.)]
Acacia nilorica
Faint traces observed in stem, roots and leaves. As-
sayed separately {Sept 1996; Xanthydrol)
Acacia obtusifolia
In some samp les as a minor component. Apparently
unpttblished work. Pers. comm. with Snu Voogelbre-
inder. Needs coufrrmation.
Appeared present in one sample ofl~avesltwigs looked
at by Mulga: Not observed in bark or root bark.
Acacia victoriae Strong blue band eo-chromatographing with 5-McO- DMT (Xanthydrol) Roots of two year old seedlings. tic by J. Appleseed 1996.
Arundo spp.
Arundo donax Leaf and flower. (Ghosal 1972a ref Trout's Notes)
=== Bromus spp. === (Brome grass)
5-MeO-DMT appeared potentially present in at least some specimens of one Bromus sp. (B. breviaristatus) 1996 tlc by J. Appleseed. Species was grown by Giorgio Samorini from seeds provided by Trout and identified at seed maturity by Dr. F. Festi in 1999. (Ref Trout's Notes)
=== Digitaria spp.=== (Crab grass)
Digitaria sanguinalis 5-MeO-DMT appeared potentially present in at least one local species; probably D. sanguinalis. Positive identification pending. 1996 tlc by J. Appleseed. (Ref Trout's Notes)
Other Grasses
??? spp. (Wild Rye, Winter Rye, Rye Grass) 5-MeO-DMT appeared to be potentially present in several local species (2-4 spp.; including both annual & perennial ryes. Including the haying material called "coastal') 1996 tlc by J. Appleseed. (Ref Trout's Notes)
Phalaris spp.
Phalaris data below is incomplete. Alkaloid concentrations and proportions are highly variable from year to year and show dramatic seasonal fluctuations. Concentrations between plant parts and first growth versus regrowih are also very different. In many populations there may be marked differences in both the amounts present and in actual alkaloid profile from one plant to the next. (i.e. plants in the same population and arising from the same seeds may show completely different chemistry, not simply differing concentrations.) See the amazing Festi & Samorini pieces listed in the Phalaris spp. page about cultivation for a review and overview of what is known so far.
- A major alkaloid in all samples they examined. (Culvenor et al. 1964)
- 5-MeO-DMT was not present in all clones examined (4 out of 12) Frahn & Illman 1973
- 5-MeO-DMT in leaf. 0.01-0.28% in material from California. (Festi & Samorini 1994a cited Welch 1971 ref Trout's Notes)
- var. AQ-1 (Italy) Weak occurrence reported (HPLC). (Festi & Samorini 1994b ref Trout's Notes)
- as Phalaris tuberosa - Strong in leaf. 2 Nov. and 17 Sept. 1995. Dec 1995. Assay. tlc by J. Appleseed 1995 (ref Trout's Notes)
- var Killer Phalaris (At one point this was synonymous with cv. Uneta, but has been in uncontrolled propagation for long enough this is no longer certain.) 5-MeO-DMT predominated in 25 June, 17 Sept., 2 Nov. 1995 samples. (DMT was predominate alkaloid in Fall 1994 by TLC) (ref Trout's Notes)
- Clone #R5 "large" amount of DMT (5-MeO? possible mistake in Trout's Notes) co-occurring with twice amount of 2-MTHBC (Clone, designated 405-9, originating with U.S. Regional Pasture Research Laboratory, University Park, Pennsylvania)
- Clone #R37 ·'trace" amount of 5-MeO-DMT co-occurring with " intermediate" amounts of hordenine and "large" amounts of 6-methoxy-2,9-dimethyl-1,2,3,4-
tetrahydro-beta-carboline. (From highly diverse source population used in plant breeding and genetic studies at the University of Minnesota. Department of Agronomy and Plant Generics".)
- Clone #R51 ·'large" amount of 5-MeO-DMT as sole observed al.kaloid. [Same source as R37)
- Clone #R96 " large" amount of 5-MeO-DMT co-occurring with "trace" amounts of hordenine and "trace" amounts of 6-methoxy-2,9-dimethyl-1,2,3,4-tetrahydro-beta-carboline. (Same source as R37) (Gander et al. 1976. ref Trout's Notes)
- cv. Australian Commercial (CPI 119305) A major alkaloid in 7 day old seedlings. 150 nmol / 100 seedlings. (Mulvena & Slaytor 1983 ref Trout's Notes); In seedlings. (Mack et al. 1988); Mature 0.05% dry wt.(Baxter & Slaytor 1972)
- cv. Sirocco 51 nmol / 100 seedlings. (Mulvena & Slaytor 1983 ref Trout's Notes); Major base. (Frahn & O'Keefe 1971); 5-MeO-DMT in leaf (Ott 1994 cited Culvenor et al 1964; Baxter & Slaytor 1972; frahn & Illman 1973 ; Moore et al 1967; Mulvena & Slaytor 1982; Oram & Williams 1967 ref Trout's Notes)
- 5-MeO-DMT in leaf and whole plant Barnes et al 1971; Culvenor eta/ 1964; Gander eta/. 1976; Majak
& Bose 1977; Majak eta!. 1978; Marten et al. \ 973; Williams et al. 197 1. Many others.
- 0.0002-0.0067% 5-MeO-DMT in material from British Columbia. (Majak & Bose 1977)
- 0-0.02% in material from Minnesota. NRG741 was strongest of those tested & NRG721 the weakest. (Majak eta/. 1978)
Festi & Samorini 1994a
- tlc by Johnny Appleseed
- P.I. 172442 Turkey (cv. Turkey Red)0.0025% to 0.045% total alkaloid by wet weight. 5-MeO is predominate alkaloid. (J.Appleseed (undated manuscript); "Ayahuasca analog plants of the temperate zone." Tlc by Johnny Appleseed: fall 1994, 25 June, 17 Sept, 2 Nov. 1995.
Extraction teks
Any typical extraction teks for DMT should potentially extract 5-MeO-DMT (but it will also extract DMT together).
Dosages and consumption methods
5-MeO-DMT is around 5 times more potent than DMT by weight.
Smoked, dosage is around 5-15mg.
Oral, according to Jonathan Ott, 30mg are active without the need for MAOIs. Mixing with MAOIs, it will be 3 times stronger, so a dosage will be around 10mg. There is some concern about 5-MeO-DMT's oral ingestion safety, and it might be related with individual metabolism differences. Check this thread for more info. If you are consuming 5-MeO-DMT orally for the first time, specially with MAOIs, start VERY low and raise your dosage gradually.
Insufflated, 5-20mg (Erowid)
History of usage
Analysis of 5-MeO-DMT
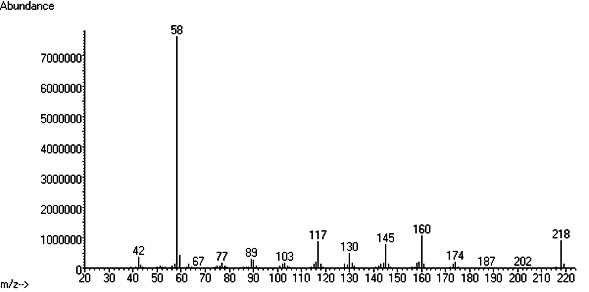
GC-MS
Retention time: 12.946 (System used)
Other info: 5-MeO-DMT: EI/MS (m/z, %): 218 (M+ , 10), 160 (6.3), 145 (2.5), 117 (5.0), 58 (100), 42 (4). (Takahashi 2008)
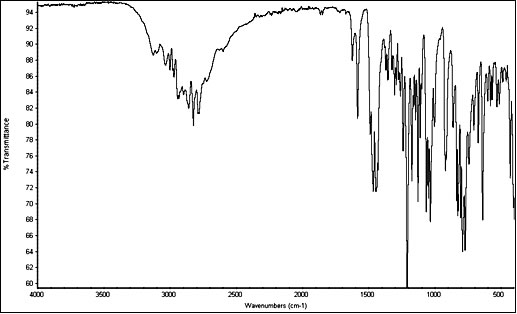
InfraRed
5-MeO-DMT freebase
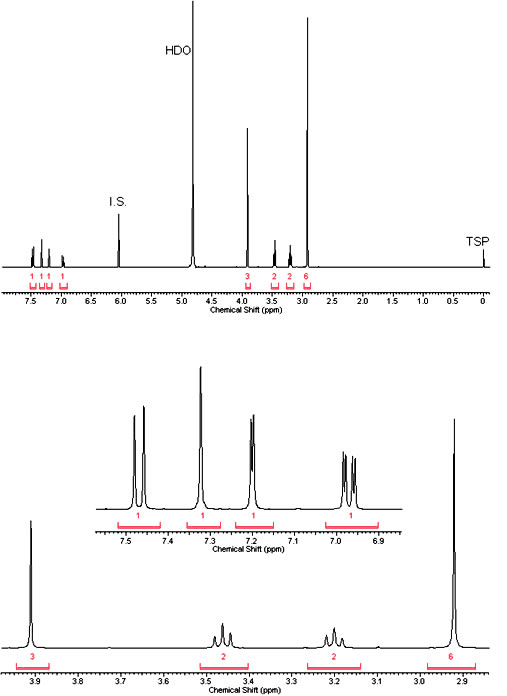
NMR
1H NMR (400 MHz, D2O) δ ppm 7.47 (d, J=8.90 Hz, 1 H) 7.32 (s, 1 H) 7.20 (d, J=2.45 Hz, 1 H) 6.97 (dd, J=8.90, 2.45 Hz, 1 H) 3.91 (s, 3 H) 3.46 (t, J=7.43 Hz, 2 H) 3.20 (t, J=7.43 Hz, 2 H) 2.92 (s, 6 H).
(Source: Microgram Bulletin Vol. 3, Pg 8)
Other info: 1 H-NMR (CDCl , δ): 2.34 (6 H, s), 2.63 (2 H, m), 2.91 (2 H, m), 3.86 (3 H, s), 6.85 (1 H, dd, J = 2.3, 8.6 Hz), 6.98 (1 H, br d, J = 2.3 Hz), 7.05 (1 H, d, J = 2.3 Hz), 7.22 (1 H, d, J = 8.6 Hz). 13 C-NMR (CDCl3 , δ): 23.7, 45.4, 56.0, 60.1, 100.8, 111.8, 112.1, 113.9, 122.3, 127.8, 131.5, 153.9. (Takahashi 2008)