Difference between revisions of "Chilled Acetone with IPA and Naphtha"
(→Paste 🌵) |
(→Paste 🌵) |
||
| Line 92: | Line 92: | ||
== Paste 🌵== | == Paste 🌵== | ||
Ethyl acetate is an ester and extracts unwanted plant esters such as chlorophyll. Fortunately, Microwave Assisted Saponification (MAS<ref>Microwave assisted saponification[https://www.mdpi.com/2304-8158/10/2/445/pdf]</ref>) breaks down plant esters. The general saponification reaction is: | Ethyl acetate is an ester and extracts unwanted plant esters such as chlorophyll. Fortunately, Microwave Assisted Saponification (MAS<ref>Microwave assisted saponification[https://www.mdpi.com/2304-8158/10/2/445/pdf]</ref>) breaks down plant esters. The general saponification reaction is: | ||
| + | |||
'''<span style="color: Orange"> <div style="text-align: center;">Ester + OH<sup>-</sup> ⇒ Carboxylate Ion<sup>-</sup> + Organic Alcohol</div></span>''' | '''<span style="color: Orange"> <div style="text-align: center;">Ester + OH<sup>-</sup> ⇒ Carboxylate Ion<sup>-</sup> + Organic Alcohol</div></span>''' | ||
Revision as of 12:03, 19 June 2021
Contents
Introduction 🙏
CIELO stands for Crystals In Ethyl-acetate Leisurely Over-the-counter.
In this technique (TEK), cold cactus alkaline paste (Fig. 2) is extracted with chilled ethyl acetate (Fig. 3). Mescaline citrate is precipitated with citric acid (Fig. 4) and collected (Fig. 5).
Thanks to everyone who contributed to this process: someblackguy, Benzyme, shroombee, Metta-Morpheus, Downwardsfromzero, Kash, grollum, Mindlusion, Doubledog, Dreamer042, Loveall, and others.
Safety ⛑️
Review ethyl acetate[1], and citric acid[2] safety information. Verify solvent MSDS, plastic compatibility, and clean evaporation.
Following this advice does not guarantee safety. It is up to each adult individual to make their own decision.
Materials 🛒
- French press (or similar ensemble)
- 300g water
- 25 of lime
- 100g dry cactus powder (outer skin or whole plant)†
- 100g anhydrous CaCl2 (ice melt)
- 1qt ethyl acetate (MEK substitute)
- Coffee filters, support basket, and funnel
- Quart jar
- 1/2 tsp Citric acid
- Washing soda and activated carbon (for solvent reclaim)
†If starting with fresh cacti, store the dark for 3 months, slice into ~half inch disks, dry in oven at lowest heat setting, and grind to a fine powder (keeping spines is ok)[3].
Process 📜
Paste 🌵
Mix water, lime, CaCl2 first, then add cactus powder. Microwave avoiding an over boil stirring frequently until 100g of water evaporates. Place in freezer (~0F) overnight (Fig. 2).
Quick and dirty option: Simply mix ice water lime and cactus powder. This simpler paste is but can work.
Extract 👨🏾🔬
Cover paste with freezer chilled ethyl acetate (~0F), mix for 60s, and filter into quart jar. Repeat until quart jar is full (~5x).
Inspect extract for droplets or particles. If present, remove them. Extract needs to be clean (see Fig. 3).
Crystalize ✨
Drop (do not stir) citric acid into extract. Clouds form quickly, slowly followed by mescaline citrate crystals (Fig. 4). Allow crystalization to complete undisturbed (~12 hours).
Note: If using whole cactus more citric acid may be needed to induce crystallization (up to 1 Tbsp).
Collect 💖
Swirl crystalized extract to knock off crystals from walls and dissolve any remaining citric acid granules. Catch floating crystals in a coffee filter. Repeat with a small amount (~1oz) of fresh room temperature ethyl acetate until all crystals are in the filter and off color is washed (~2-3x). Dry and collect from filter (Fig. 5).
Yield depends on the cactus and is usually between 0.2% to 2% with ~1% being common[4].
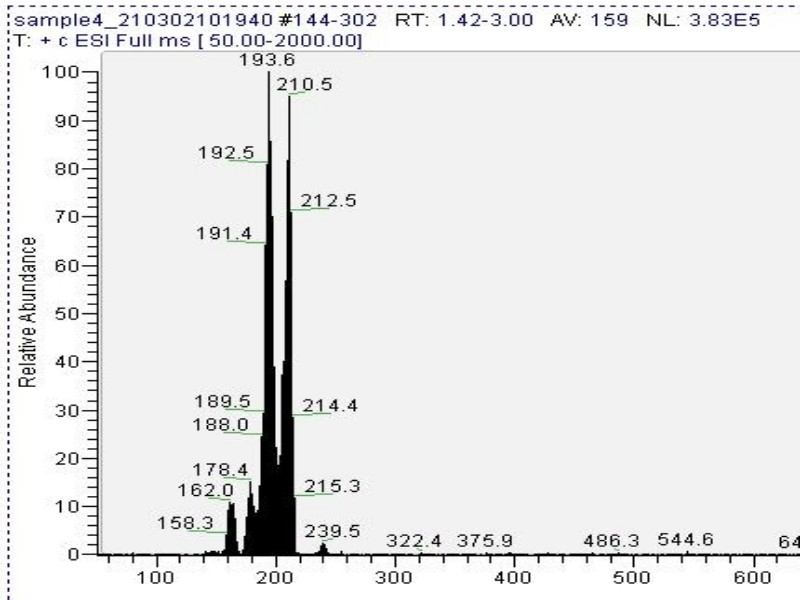
Mass spectrometry (MS) results from solaris analytical[5] indicate the product is very clean mescaline (Fig. 6).
Reclaim Solvent 💚
Reusing solvents is encouraged[6] at the DMT nexus.
Wash extract with sodium carbonate saturated water shaking vigorously (emulsions do not form). Filter any excess calcium carbonate and remove water layer. Freeze and filter out ice crystals. If a yellow color develops after multiple reuses it can be removed by resting over activated carbon for several days.
Appendix: Development Notes 🔬
Paste 🌵
Ethyl acetate is an ester and extracts unwanted plant esters such as chlorophyll. Fortunately, Microwave Assisted Saponification (MAS[7]) breaks down plant esters. The general saponification reaction is:
An important example is,
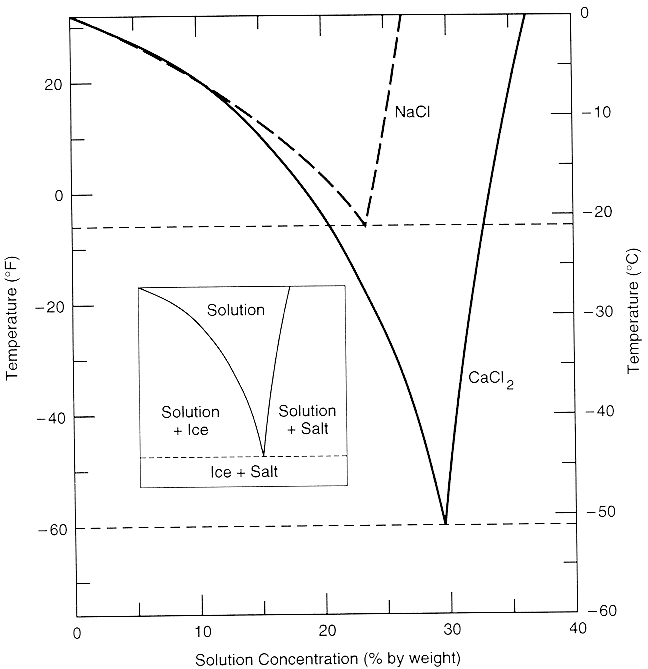
On the other hand, adding CaCl2 to ~27% enables extraction below ~0F (see Fig. 7). Cold extractions at ~0F do not dissolve large organic plant molecules well, while mescaline solubility remains favorable.
MAS and cold extraction combine to significantly reduce the amount of plant matter in the extract, helping crystallization and reducing contaminants.
In other experiments, sodium bases and salts (Sodium carbonate, sodium hydroxide, and sodium chloride) where tested but made the paste congeal lowering yields.
Extract 👨🏾🔬
Longer/warmer pulls resulted in darker extract, smaller crystals, solvent paste absorption, congealing of paste, and no yield benefit.
Chemically drying the extract had no benefits.
An additional long room temperature pull on the TEK's spent paste only yielded 4mg of very small crystals, indicating the chilled pulls are efficient.
Crystalize ✨
During crystallization, every 233mg of citric acid (H3Cit) react with free base mescaline (Mes) to form to 1g of mescaline citrate (or slightly more if a hydrate is precipitating):
Excess citric acid shifts the precipitation reaction to the right (Le Chatelier's principle), helping overcome water and plant material. There is a lot room for excess citric acid in solution since its solubility is 50mg/g in ethyl acetate. The TEK guidelines range from 1.5mg/ml to 20mg/ml, but since cacti and pull techniques can vary, users may find other values work better for their specific situation (in one specific example with whole cactus powder 20mg/g was used [8]).
Several factors can make crystals smaller: Reusing ethyl acetate, longer/warmer pulls, higher citric acid concentration, mechanical agitation, and other potential variables. Small crystals can look like a fine powder. Potency does not seem affected by the crystallization appearance, and a powdery precipitate is not a problem unless it becomes difficult to decant/filter.
After the initial crystallization, adding more citric acid and/or moving the extract to the refrigerator did not result in any more precipitation. Moving the extract to the freezer produced ice crystals.
Other dry organic acids could work. Fumaric, Malic, Tartaric, Ascorbic, Succinic, etc can be tested in future investigations.
Collect 💖
Washing crystals in a filter appears to wick away plant colors and superior to decanting. Unlike the pulls, warmer ethyl acetate is preferred to wash off plant matter.
The quart jar wash should be done immediately. If any straggler crystals dry in the jar they may stick to the wall. To recover from such a situation, dissolve stuck crystals in hot water, dry in a shallow dish, and scrape.