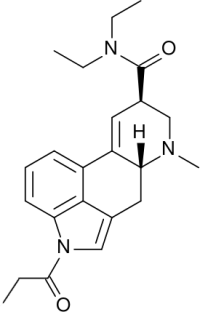
1P-LSD
| Safety profile | |
|---|---|
Category:
|
|
Main effects:
|
|
Side effects:
|
|
Health risks:
|
|
Important safety remarks:
|
|
Chemical and physical properties
- Systematic (IUPAC) name: (6aR,9R)-4-propionyl-N,N-diethyl-7-methyl-4,6,6a,7,8,9-hexahydroindolo[4,3-fg]quinoline-9-carboxamide
- Synonym: 1-Propionyl-lysergic acid diethylamide
From an old Wikipedia draft:[2]
Sharing a strong structural similarity with ALD-52, the chemical purportedly used in the 1960's Orange Sunshine 1P-LSD follows the less common path of modifying the 1- position on the LSD molecule which in the past has been avoided due to unpredictable results or lack of activity.[3] Lots of debate is surrounding the action of 1P-LSD and is unclear exactly how the chemical is functional with an unconfirmed quote from David E. Nichols believing the chemical should be inactive while most other speculation believing it would simply follow the same metabolic path as ALD-52 and undergo conversion in vivo to LSD.
"I am sure that the 1-propionyl would also hydrolyze off of an indole, but I don't know whether in vivo conditions would work. In a chemistry lab, you can get off an N-benzoyl, so an N-propionyl will probably come off too. But in the body? I don't know the answer to that. The compound would not be active as the N-propionyl however. Way that LSD docks into the 5-HT2A receptor, the indole NH hydrogen bonds to serine 5.46. With the propionyl, it won't fit into the receptor."[4]
Links
- bluelight.org: The Big & Dandy 1P-LSD (1-propionyl-lysergic acid diethylamide) Thread
- reddit.com: New Novel Lysergamide 1P-LSD
- https://en.wikipedia.org/w/index.php?title=Draft:1P-LSD&oldid=647406505
References
- ↑ bluelight.org: The Big & Dandy 1P-LSD (1-propionyl-lysergic acid diethylamide) Thread
- ↑ https://en.wikipedia.org/w/index.php?title=Draft:1P-LSD&oldid=647406505
- ↑ http://chemistry.mdma.ch/hiveboard/palladium/pdf/Ergot%20-%20The%20Genus%20Claviceps%20(1999)/TF3168ch8.pdf
- ↑ http://www.shroomery.org/forums/showflat.php/Number/21106929#21106929