Difference between revisions of "Chilled Acetone with IPA and Naphtha"
(→Strategy ♟️) |
(→Strategy ♟️) |
||
| Line 138: | Line 138: | ||
| − | Fortunately, DMT aggregates can break down in acidic conditions. Therefore, to simultaneously break down DMT aggregates and plant material, a long acidic pressure cooking step is used (described before by for example Northener). Vitamin C is chosen as the source of acid due to its good experimental performance, but other acids could also work. Subsequently, relatively gentle ionic strength (no added salt), gentle alkaline pH (no excess lye beyond emulsion breakdown), and low DMT concentration (<0.5%) conditions are used to minimize any DMT re- | + | Fortunately, DMT aggregates can break down in acidic conditions. Therefore, to simultaneously break down DMT aggregates and plant material, a long acidic pressure cooking step is used (described before by for example Northener). Vitamin C is chosen as the source of acid due to its good experimental performance, but other acids could also work. Subsequently, relatively gentle ionic strength (no added salt), gentle alkaline pH (no excess lye beyond emulsion breakdown), and low DMT concentration (<0.5%) conditions are used to minimize any DMT re-polymerization. Naphtha is introduced before the basing step to minimize the time bulk DMT spends in alkaline water as the pH initially increases. |
== Vitamin C 🍊== | == Vitamin C 🍊== | ||
Revision as of 18:18, 20 October 2021
Contents
Introduction 🙏
In this technique (TEK), potential DMT polymer (also called goo, aggregate, or oligomer) is minimized by (1) pressure cooking with Vitamin C, and (2) using gentle alkaline conditions. By minimizing DMT polymer, a high yielding white crystalize powder is obtained directly from the plant material.
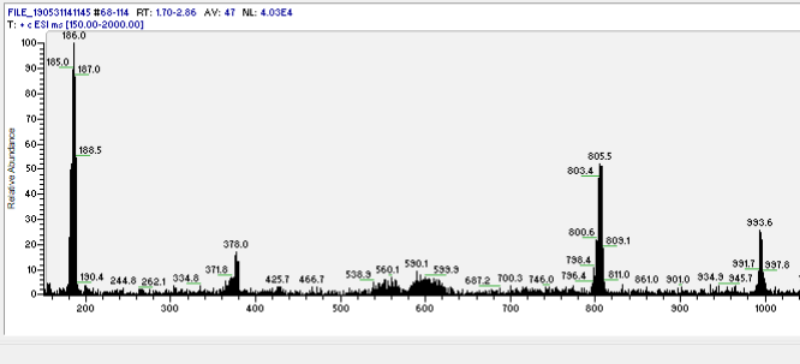
Thanks to Northerner for sharing his excellent pressure cooking experimental result, to Benzyme for showing MS evidence of DMT polymers[1] (see Fig. 1 below), and to Jees, downwardsfromzero, IridiumAndLace, and Loveall for their contributions to this process in the forum.
Safety ⛑️
Review NaOH[2] and naphtha [3] safety information. Verify solvent MSDS purity, plastic compatibility, and clean evaporation.
Never have solvents near an open flame.
Following this advice does not guarantee safety. It is up to each adult individual to make their own decision.
Materials🛒
Consumables👩🌾
- 25g ascorbic acid (vitamin C)
- 50g of mimosa hostlis root bark
- 2000ml of water
- ~250ml naphtha
- 25g of NaOH
Equipment🏺
- Quart and pint jars
- Scale
- Pressure cooker
- Pipette
- Freezer
- Fan
- Scraping tool
Process Overview 👀
- Pressure cook root bark with vitamin C
- Add naphtha and then basify
- Pull, freeze precipate, and collect
Detailed Process 📜
Pressure Cook 🔥💨
Pressure cook root bark with water and vitamin C for 4 hours. Reduce volume to <800ml and transfer to a quart jar (bark and all, do not filter). Rinse pot once with a small amount of water and add the wash to the jar.
Pull 👩🔬
Allow jar to cool to room temperature. Shake in ~85ml of naphtha (almost completely filling the jar). Add lye and shake vigorously for a few minutes. Solution will warm up slightly as lye dissolves and will quickly go from red, to milky, to dark red.
Rest solution until naphtha layer separates (~15 minutes, see Fig. 3). If separation is not complete, mix in another 5g of lye and try again.
Remove naphtha with a pipette into a pint jar. It is ok if a few drops of watery extract or bark particles come through (they will be decanted in the next section).
Add another ~80ml of naphtha to the quart jar. Shake for a few minutes, rest until layers separate, and pipette naphtha into the second jar. Perform this step one more time (total of 3 pulls, including the first one).
Ideally, all three pulls are be done within an hour while the quart jar is slightly warm from the lye dissolving in water.
Crystalize ✨
Carefully decant naphtha in pint jar to a fresh pint jar. Do not allow any watery extract or particles to come through.
If using new naphtha, one option is to evaporate the solvent until slightly cloudy with the help of a fan in a well ventilated area. Note that minimum polymer is very soluble in naphtha and tends to cloud less than other TEKs. It is OK if no cloudiness is present after 50% naphtha has evaporated, and the extract can be moved to the freezer at that point.
A better option is to skip the solvent evaporation. Yield will be lower if using new naphtha, but it will be available for reuse as a one-time "investment". Subsequently, used naphtha does not need to be evaporated before freezing to get the full yield since it already comes preloaded with a DMT concentration that is saturated at the freezing temperature.
Place second pint jar in the freezer for at least 24 hours to precipitate DMT. Decant naphtha off crystals, and immediately dry with the help of a fan. Once dry, scrape up off-white xtals. This is the final product.
Reclaim Solvent 💚
Reusing solvents is encouraged[4] at the DMT nexus.
Simply reuse freeze precipitated naphtha as-is. Re-used naphtha is saturated with DMT at freezer temperature and pre-freezer evaporation is not needed. Easy 😊
Frequently Asked Questions ❓
Q: That's a lot of hypothesis you got down in the appendix. Have any experimental evidence consistent with them?
A: Yes. Benzyme's MS, together with polymerization and de-polymerization experiments. As far as we know experiments are consistent with the hypotheses listed. The community is welcome to update this Wiki entry as more evidence arises, especially if any of the hypotheses are disproved (thank you).
Q: What's so special about Vitamin C?
A: See the development notes in appendix below.
Q: Why are there only 3 pulls without a warm water bath or salting out ionic strength? Usually ~5 warm + high ionic strength pulls are needed (40-50C)
A: DMT monomer is highly soluble in naphtha and has an excellent partition coefficient. By converting natural DMT to this form, and keeping alkaline conditions gentle to avoid polymerization, the pulls are simpler and very efficient.
Appendix: Development Notes 🔬
Hypotheses 🤔
This TEK hypothesizes that:
- Not all of the DMT is in the plant in monomer form, some of it is in macro-molecule form (also called polymer, oligomer, or goo)
- In addition to natural DMT polymer, even more polymer can form during the basing step under high alkaline, high ionic strength, and high DMT concentration conditions
- A pressure cooker treatment of the bark using vitamin C is effective at breaking down any natural DMT polymer to the monomer form
- Once natural DMT polymer is broken down, gentle alkaline conditions keep it from forming again
- DMT monomer properties compared to DMT polymer:
- Easier to dissolve in naphtha (better partition coefficient)
- Clouds later during naphtha evaporation
- Slowly crashes during freeze precipitation as white crystals. In contrast, DMT polymer precipitates sooner as yellow/orange/red semisolid goo
- Easier to handle and dose precisely
- Low and consistent vaporization temperature, ideal for newer electronic vaporization devices with precisely tuned temperature settings
- Visibly unique upon crystalization, eliminating questions around plant oil contaminants
- May be easier to complex with HPBCD for sublingual administration
- It is unknown if it has better bioavailability for oral or rectal administration. In principle, stomach acid should be able to break down DMT polymer, so perhaps there is no difference for oral administration
- There is no expected benefit for torch vaporization by an experienced user since the strong heat produced manually can easily vaporize everything. However, the process window between vaporizing and burning the DMT is larger for the monomer which may benefit the inexperienced user
Strategy ♟️
The strategy of this TEK is to break down both DMT aggregates and plant material, while minimizing DMT re-aggregation during the basing step.
Agressive alkaline concentration conditions are avoided. While these type of processes can break down plant material, their downside is that they don't break down natural DMT aggregates and can even increase the degree of polymerization.
Fortunately, DMT aggregates can break down in acidic conditions. Therefore, to simultaneously break down DMT aggregates and plant material, a long acidic pressure cooking step is used (described before by for example Northener). Vitamin C is chosen as the source of acid due to its good experimental performance, but other acids could also work. Subsequently, relatively gentle ionic strength (no added salt), gentle alkaline pH (no excess lye beyond emulsion breakdown), and low DMT concentration (<0.5%) conditions are used to minimize any DMT re-polymerization. Naphtha is introduced before the basing step to minimize the time bulk DMT spends in alkaline water as the pH initially increases.
Vitamin C 🍊
Experimentally, Vitamin C produced better results compared to acetic and citric acids. Vitamin C is biologically active as a mild antioxidant and reducing agent and can pass through cell membranes. During pressure cooking, Vitamin C breaks down into several other acids such as dehydroascorbic acid, diketogulonic acid, xylonic acid, threonic acid and oxalic acid. It could be that by being exposed to multiple different acids DMT de-polymerization is improved by complimentary mechanisms.
Other acids may also work, and the kitchen alchemist is encouraged to report on any new experimental results (both positive and negative).
Cloudiness 🌫️
DMT monomer does not readily form clouds in naphtha. Compared to other extractions that do not minimize polymer, clouds form later in the evaporation process and are not as opaque. This is a good sign and not a cause for concern. It is OK to freeze precipate before clouds are observed after reducing the solvent volume by ~50% or more. Re-using naphtha avoids this complication since it is already saturated with DMT at the freezer temperature (no naphtha evaporation is needed).